Clinical Research Coordinator Resume Examples, Templates & Writing Guide

Written by: Scale.jobs EditorialLast updated: May 1, 2026








Introduction
Build a clinical research coordinator resume that showcases IRB protocol management, patient enrollment expertise, GCP compliance, and data integrity skills for academic medical centers and CRO positions.
This guide walks you through every major section of a clinical research coordinator resume, with practical tips you can apply today.
- How to present clinical trial management experience and enrollment metrics effectively
- Strategies for highlighting GCP certification, IRB submission expertise, and regulatory compliance
- Techniques for showcasing CTMS and EDC platform proficiency on your research resume
- Methods for quantifying study enrollment rates and protocol adherence outcomes
- How to demonstrate informed consent processes and participant safety monitoring skills
- Key approaches for tailoring your resume to academic, pharmaceutical, or CRO settings
Clinical Research Coordinator resume guide
Below, you will find section-by-section guidance for your clinical research coordinator resume — from your opening summary through skills and experience. Tailor every line to the job you want.
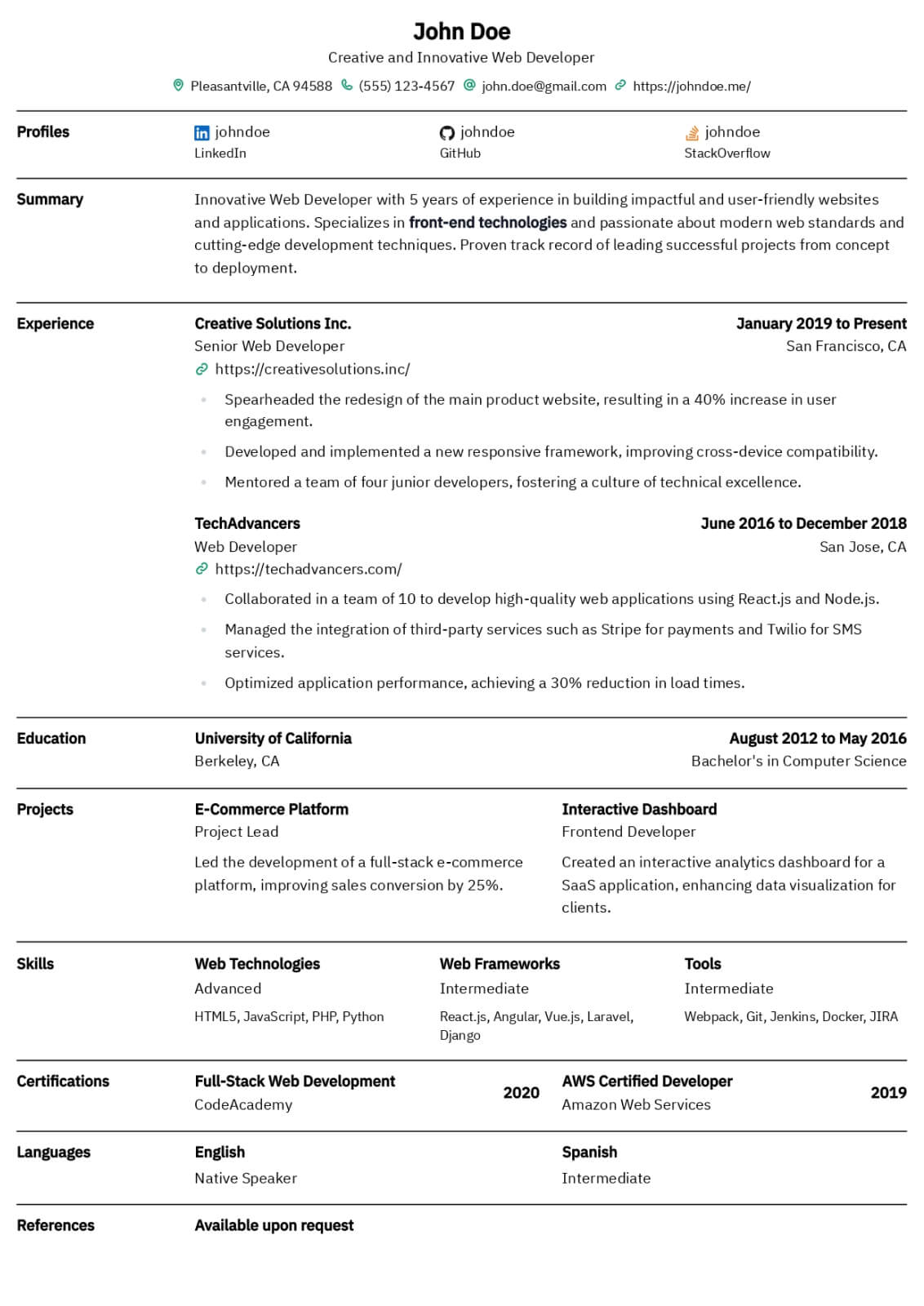
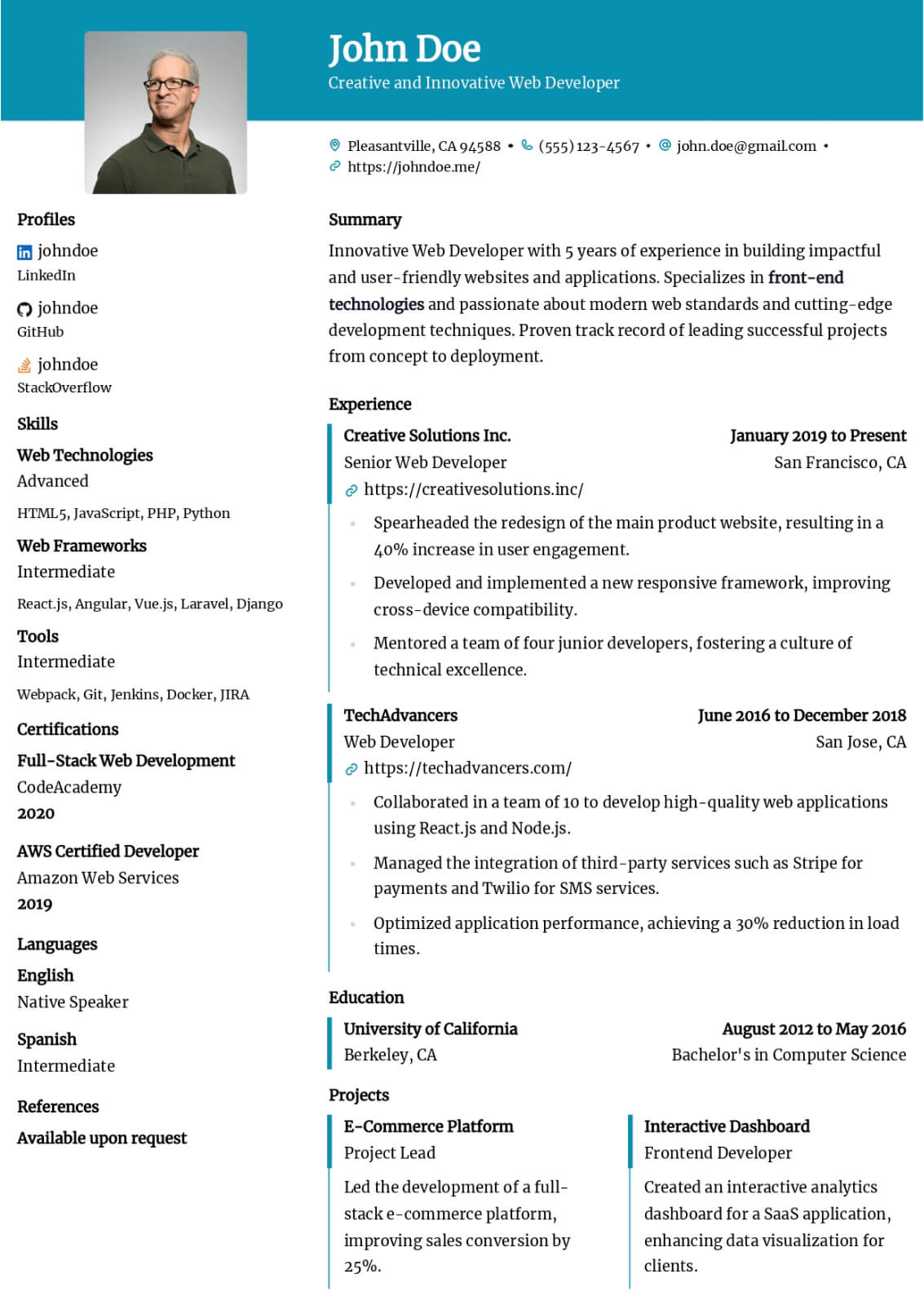
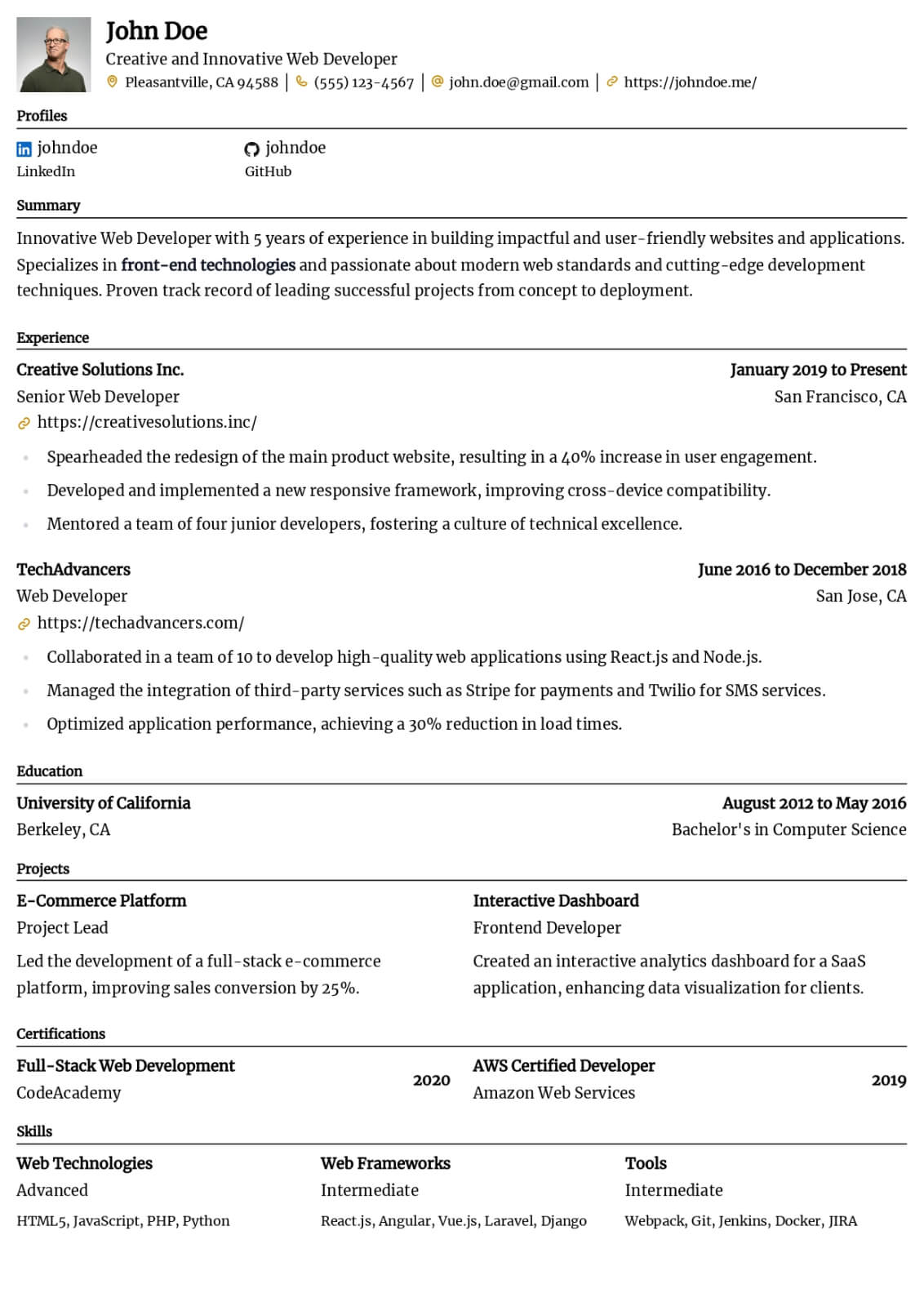
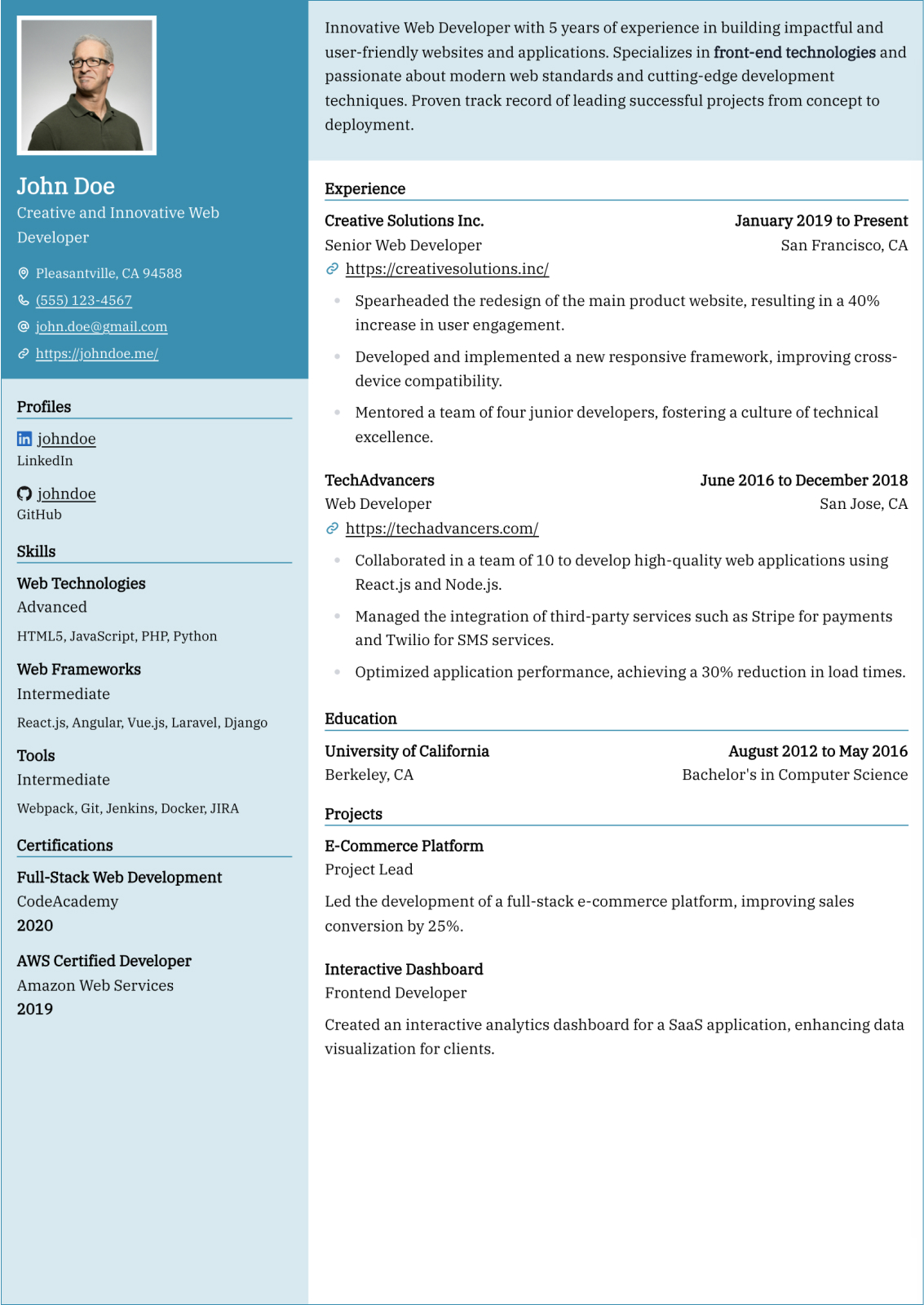
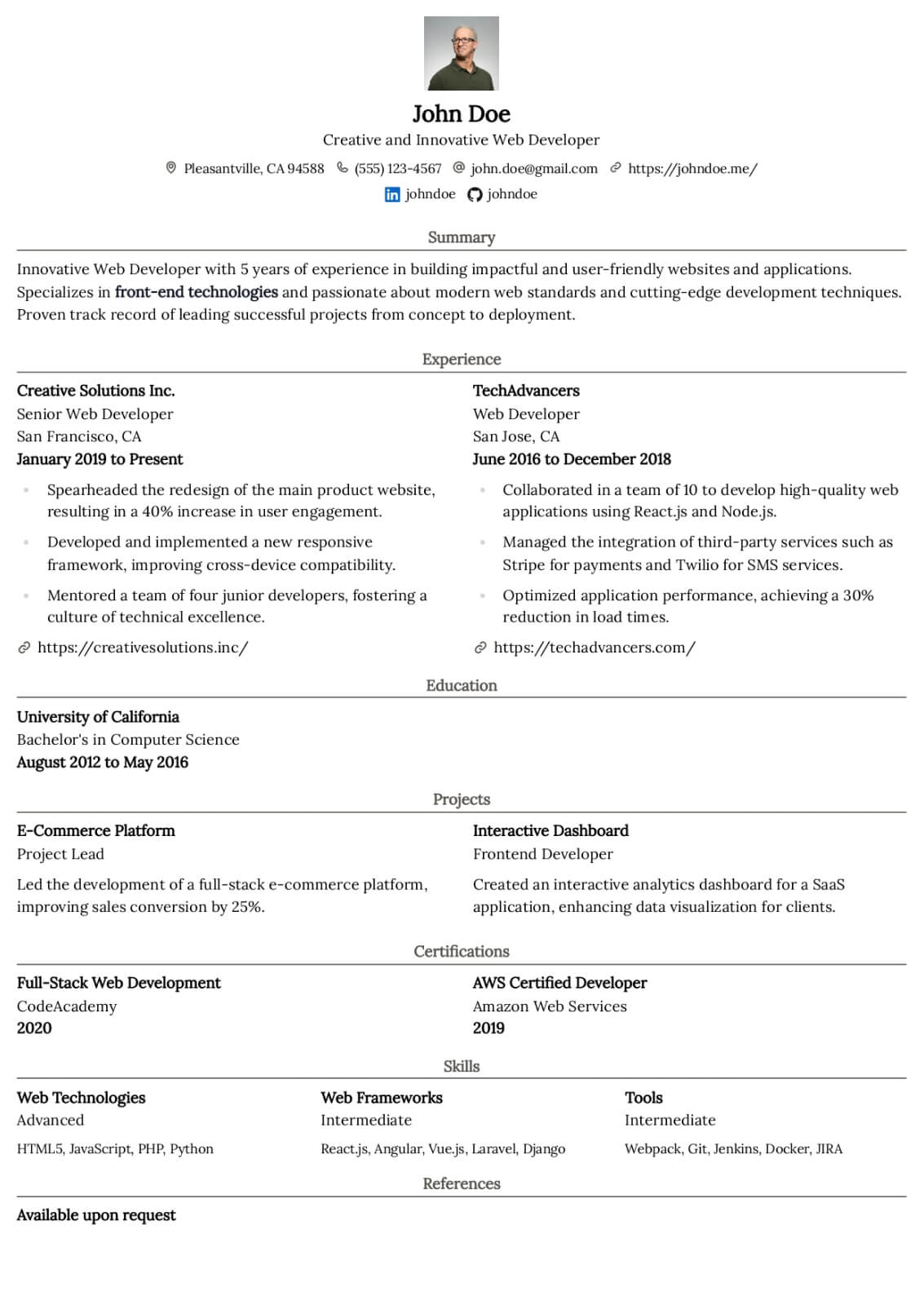
Professional Summary
Your professional summary should position you as an experienced clinical research coordinator with demonstrable protocol management skills and regulatory compliance expertise. Open by stating your GCP certification status, years of clinical research experience, and primary therapeutic area such as oncology, cardiology, or neurology. Reference the number and phase of clinical trials you have coordinated, for example managing five concurrent Phase II and Phase III oncology trials across a multi-site academic medical center. Include a quantified enrollment metric such as achieving one hundred fifteen percent of target enrollment in a pivotal Phase III study within the original recruitment timeline. Mention your proficiency with clinical trial management systems and electronic data capture platforms. Keep the summary between three and five sentences for clarity.
Research Experience
Present your clinical research positions in reverse-chronological order, listing the institution or CRO name, therapeutic area, and employment dates. Write four to six bullet points per role using action verbs such as coordinated, consented, monitored, submitted, and documented. Quantify your impact by noting the number of active studies managed, participant enrollment numbers, screen failure rates, and protocol deviation frequencies. Describe specific responsibilities including managing IRB submissions and continuing reviews, conducting informed consent processes, performing protocol-required assessments and specimen collection, and maintaining regulatory binders. Highlight sponsor and monitor interactions by describing how you prepared for monitoring visits and resolved queries. Include experience with serious adverse event reporting, data safety monitoring board coordination, or study closeout activities to demonstrate full lifecycle trial management capability.
Research & Regulatory Skills
Build a skills section with eight to ten research-specific hard skills and six to seven professional soft skills. Hard skills should include ICH-GCP guideline compliance, IRB submission and protocol amendment management, informed consent documentation, clinical trial management system proficiency with platforms like Medidata Rave, REDCap, Oracle Clinical, or Veeva Vault, and electronic data capture system navigation. Add skills in regulatory document preparation, serious adverse event reporting, specimen processing and biobanking, and FDA audit readiness. Soft skills should emphasize participant rapport and retention strategies, investigator and sponsor relationship management, meticulous documentation habits, and ability to manage multiple concurrent protocols under competing deadlines. Use clinical research terminology to ensure ATS recognition by academic medical centers and contract research organizations.
Certifications & Training
Lead with your GCP certification from a recognized provider such as CITI Program, TransCelerate, or NCI as this is the foundational compliance credential for clinical research professionals. Include your Certified Clinical Research Coordinator credential from ACRP or Certified Clinical Research Professional from SOCRA if you hold these industry certifications. List any CITI Program human subjects protection training, HIPAA research certification, and protocol-specific training completions. Add BLS certification if your role involves direct patient contact during study visits. If you are pursuing clinical research certification, include the expected examination date to demonstrate professional development commitment. Place this section prominently because research sponsors and institutions verify GCP training status during study startup.
Education
List your bachelor's or master's degree in a relevant field such as nursing, biology, public health, or clinical research administration along with the institution name and graduation year. Clinical research coordinator roles typically require a minimum of a bachelor's degree in a health-related field. Include relevant coursework in biostatistics, epidemiology, research methodology, or regulatory affairs that supports your research competency. Mention any clinical research certificate programs or graduate-level training in clinical trial management that supplements your degree. If you hold an RN license or other clinical credentials that enhance your research qualifications, reference them here to demonstrate the clinical foundation supporting your research expertise.
Publications & Presentations
Include a section for peer-reviewed publications, conference presentations, and scholarly contributions if you have authored or co-authored research outputs. List publications in standard citation format including all authors, article title, journal name, and publication year. Reference poster or podium presentations at professional conferences such as ACRP, SOCRA, or disease-specific meetings like ASCO or AHA. Describe any contributions to study protocols, investigator brochures, or standard operating procedure development. These entries are particularly valuable for candidates targeting academic medical center positions where research productivity is valued alongside clinical trial coordination skills and contributes to your professional profile.
Resume layout and formatting
Use a clean, single-column layout with clear section headings and plenty of white space. Lead with technical strengths such as ICH-GCP Guideline Compliance, IRB Submission & Protocol Amendment Management, Informed Consent Process & Documentation, CTMS Proficiency (Medidata Rave, REDCap, Veeva Vault), Electronic Data Capture (EDC) Navigation, Serious Adverse Event (SAE) Reporting, then reinforce interpersonal strengths like Participant Rapport & Retention, Investigator & Sponsor Relationship Management, Meticulous Regulatory Documentation, Multi-Protocol Time Management. Keep fonts standard (e.g., Arial or Calibri) at 10–12pt body size so your resume stays ATS-friendly and easy to scan.
Key takeaways
- Lead with GCP certification and ACRP or SOCRA credentials prominently displayed
- Quantify enrollment metrics and protocol adherence rates with specific study outcomes
- Name CTMS and EDC platforms to match employer or sponsor technology requirements
- Highlight therapeutic area expertise that aligns with the target research program
- Include regulatory compliance experience such as IRB submissions and audit preparation
- Demonstrate full trial lifecycle management from startup through closeout activities
Build your Clinical Research Coordinator resume with Scale
Lead with GCP certification and ACRP or SOCRA credentials prominently displayed
Use This Template
Professional Templates That Make You Stand Out
Browse modern, ATS-friendly resume designs crafted to impress recruiters. Customize any template and download it as a Word or PDF file.
Listen What Our Users Have to Say
Rohan Sen
I am very happy with the team's quick turnaround time - any query is responded at utmost priority. Shoutout to my client manager, Anub Biju - very helpful.
Gael L
Service and communication is great, cover letters are non-ai sounding and well tailored. Just have a lot of communication and review with your staff!
Jonathan Parry
Wow - don't tell your peers! Wow, I can't recommend scale.jobs enough - it's so good I am not sharing with my peers. Applications at scale that get through filters. Thank you!
Cynthia Zhu
Great service! The scale.jobs team was very responsible and managed to apply tons of jobs for me in a very tight deadline to help me secure interviews quickly. Highly recommend to anyone who needs help applying to jobs!
Yash Yenugu
Save your fingers. Saved me from a thumb cramp because we're expected to effortlessly apply to jobs during these times.
Cian O'Driscoll
Clever service. Takes the hard effort out of applying for jobs with an intuitive dashboard and attention to detail. A great asset to job seekers. :-)
Frequently asked questions
What should a clinical research coordinator resume include in 2026?
A strong CRC resume should feature GCP certification, a professional summary with enrollment metrics, detailed research experience organized by therapeutic area and trial phase, CTMS and EDC platform proficiency, and relevant education. Include regulatory compliance skills such as IRB management and SAE reporting. Tailor the resume to the target institution's therapeutic focus and research portfolio.
Is GCP certification required for clinical research coordinator positions?
GCP certification is required by virtually all research sponsors and institutional employers. It demonstrates your understanding of ICH-GCP guidelines governing human subjects research conduct. Most institutions require GCP training completion before you can be listed on study delegation logs or interact with research participants. Maintaining current certification is essential for ongoing eligibility.
How do I quantify research experience on a CRC resume?
Reference specific metrics including the number of concurrent studies managed, trial phases coordinated, total participants enrolled, screen failure and retention rates, and protocol deviation frequencies. Include enrollment achievement percentages such as reaching one hundred ten percent of target enrollment. These numbers demonstrate your operational effectiveness and are the primary criteria research hiring managers evaluate.
What CTMS and EDC platforms should I list on my resume?
List every clinical trial management and data capture platform you have used including Medidata Rave, REDCap, Oracle Clinical, Veeva Vault, OnCore, and Florence eBinders. Describe your proficiency level and the functions you performed within each system. Platform experience is a critical hiring factor because sponsor-mandated systems vary by study and training time affects study startup timelines.
How long should a clinical research coordinator resume be?
Most CRCs benefit from a one to two page resume. New coordinators with fewer than three years of experience should aim for one page. Experienced coordinators managing multiple therapeutic areas with publications and certifications can appropriately use two pages. Ensure every entry demonstrates research competency and regulatory compliance relevant to the target position.
How do I transition from clinical nursing to clinical research coordination?
Emphasize transferable clinical skills such as patient assessment, informed consent experience, specimen collection, adverse event monitoring, and clinical documentation. Highlight your understanding of medical terminology, disease processes, and patient communication from your nursing background. Obtain GCP certification and consider ACRP or SOCRA credentials to validate your research competency before applying.
Related Resume Guides
Registered Nurse
Craft a compelling registered nurse resume that highlights clinical competencies, patient care outcomes, EMR proficiency, and licensure credentials to land interviews at top healthcare facilities.
Nurse Practitioner
Build a polished nurse practitioner resume that showcases autonomous prescriptive authority, diagnostic expertise, patient panel management, and advanced practice licensure for competitive APRN positions.
Health Information Technician
Craft a health information technician resume that highlights EHR data management expertise, RHIT credentials, HIPAA compliance knowledge, and clinical data integrity skills for HIM department roles.
Medical Coder
Build a medical coder resume that demonstrates ICD-10-CM/PCS mastery, CPT coding accuracy, CPC or CCS certification, and revenue cycle impact to secure positions in hospitals, clinics, and remote coding organizations.
Physician Assistant
Create a physician assistant resume that highlights diagnostic acumen, procedural competence, PA-C certification, and collaborative practice experience to secure positions in hospitals, clinics, and surgical teams.